Notes of chemistry
Here we are providing the full length Chemistry Notes free of cost. These notes are important for class 12th (all boards). even these are important for various competitive exam like PGT, TGT, PCS,NEET,JEE exams.
Definitions
Binary solution: A solution having two components is called a binary solution. Components of a binary solution are solute and solvent. The component that is having a greater number of moles is known as solvent. Solvent determines the physical state of the solution.
1. When the solvent is in solid state, solution is called Solid solution.
2. When the solvent is in liquid state, solution is called Liquid solution.
3. When the solvent is in gaseous state, solution is called Gaseous solution.
Water is a universal solvent because of its high dielectric constant (approximately 82)
| SOLUTE | SOLVENT | TYPES OF SOLUTION | EXAMPLE |
| Solid | Solid | Solid Solution | Alloy |
| Liquid | Solid | Solid Solution | Amalgams |
| Gas | Solid | Solid Solution | Gas adsorbed on solid surface |
| Solid | Liquid | Liquid Solution | Sugar or salt solution in water |
| Liquid | Liquid | Liquid Solution | Alcohols in water, Dilute acids |
| Gas | Liquid | Liquid Solution | CO2 or O2 in water |
| Solid | Gas | Gas Solution | Dust or smoke in air |
| Liquid | Gas | Gas Solution | Water vapours in air, chloroform in nitrogen gas |
| Gas | Gas | Gas Solution | Air |
Saturated solution
A solution is said to be saturated if it holds the maximum amount of solute at a given temperature in a given quantity of the solvent.
Solubility
It may be defined as the maximum amount of solute that can be dissolved in 100 g of solvent at a specified temperature. The solubility of solid into liquid depends upon the following factors:
(i) Nature of solute (ii) Nature of solvent (iii) Temperature
Solubility of solids increases on increasing the temperature whereas solubility of gases decreases on increasing the temperature.
Concentration Terms

Some Important Relations

Henry’s Law
It states that at a constant temperature, the solubility of a gas in a liquid is directly proportional to the pressure of the gas.
The most commonly used form of Henry’s law states that the partial pressure (p) of a gas in vapours phase is proportional to the mole fraction of the gas (x) in the solution and is expressed as P = KH . X
Here KH is the Henry’s law constant and X is the mole fraction of the gas. As the temperature increases Henry’s law constant, KH increases so the solubility of the gas in the liquid decreases.
Applications of Henry’s Law: –
1. To increase the solubility of CO2 in soda water and soft drinks, the bottle is sealed under high pressure.
2. To avoid the toxic effects of high concentration of nitrogen in the blood, the tanks used by scuba divers are filled with air diluted with helium (11.7% helium, 56.2% nitrogen and 32.1% oxygen).
3. At high altitudes, low blood oxygen causes climbers to become weak and make them unable to think clearly, which are symptoms of a condition known as anoxia.
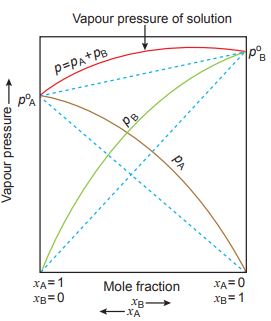
Raoult’s Law
According to Raoult’s law for a solution of volatile liquids the partial vapour pressure of each component in the solution is directly proportional to its mole fraction.
PA ∝ XA Similarly PB ∝ XB
PA = PoA . XA Similarly PB = PoB . XB.
For Vapor phase:- If yA and yB are the mole fractions of components A and B respectively in the vapour phase, then
pA = yA . Ptotal
pB = yB . Ptotal
Raoult’s law becomes a special case of Henry’s law in which KH becomes equal to POA
Raoult’s law for non-volatile solute
For a solution containing non-volatile solute present in a volatile solvent, Raoult’s law may be stated as the relative lowering of vapour pressure for a solution is equal to the mole fraction of solute.
Relative Lowering in vapour pressure is equal to mole fraction of non volatile solute.
(Po – P) / Po = XB
where Po is the vapour pressure of pure solvent, P is the vapour pressure of solution, XB is the mole fraction of non volatile solute.
Ideal and Non Ideal solutions
| Ideal Solutions | Non Ideal solution |
| The solutions that obey Raoult’s Law over the entire range of concentrations are known as ideal solutions. | When a solution does not obey Raoult’s Law over the entire range of concentration, then it is called non-ideal solution. |
| dHmix =0 and dVmix =0 | Change in enthalpy (dH) on mixing is not equal to 0, and dV mixing is not equal to 0 |
| The intermolecular attractive forces between solute molecules and solvent molecules are nearly equal to those present between solute and solvent molecules i.e. A-A and B-B interactions are nearly equal to those between A-B. | The intermolecular attractive forces between solute molecules and solvent molecules are not equal to those present between solute and solvent molecules i.e. A-A and B-B interactions are not equal to those between A-B. |
| Example:- Mixture of methanol and ethanol, Mixture of n-hexane and n-heptane, Mixture of benzene and toluene. | Example:- Mixture of Carbon tetrachloride and benzene, Mixture of Acetone and benzene, Mixture of Acetic acid and pyridine, Mixture of Hydrochloric acid and water |
From solid State check these notes also
Solid State full length notes
Solid State MCQ with solved pdf
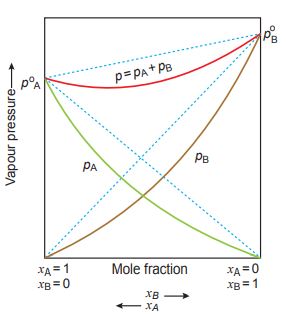
Deviation from Raoult’s Law
| Positive deviation | Negative Deviation |
| The partial pressure of each component and the resultant total pressure are greater than the pressure expected on the basis of Raoult’s law. pA > pAo XA ; pB > pBo XB | The partial pressure of each component (A and B) and resultant total vapour pressure are less than the pressure expected on the basis of Raoult’s law. pA < pAo XA ; pB < pBo XB |
| dHmix > 0, dVmix > 0 | dHmix < 0, dVmix < 0 |
| Intermolecular forces between solvent–solute molecules (A – B) are weaker than those between solvent–solvent (A – A) and solute–solute (B – B) molecules | Intermolecular forces between solvent–solute molecules (A – B) are stronger than those between solvent–solvent (A – A) and solute–solute (B – B) molecules |
| (i) Ethyl alcohol and water (ii) Acetone and carbon disulphide (iii) Carbon tetrachloride and benzene (iv) Acetone and benzene (v) Ethanol and CCl4 (vi) Benzene and methanol (vi) Chloroform and water (vii) Methanol and chloroform (viii) Acetic acid and toluene | Example:- (i) HNO3 and water (ii) Chloroform and acetone (iii) Acetic acid and pyridine (iv) Hydrochloric acid and water (v) Chloroform and methyl acetate (vi) Chloroform and benzene |
 |  |
Azeotropes
Azeotropes are binary mixtures having same composition in liquid and vapour phase and boil at constant temperature like a pure solvent. Liquids forming azeotrope cannot be separated by fractional distillation. There are two types of azeotropes called minimum boiling azeotrope and maximum boiling azeotrope.
Minimum boiling azeotrope
The solutions which show a large positive deviation from Raoult’s law form minimum boiling azeotrope at a specific composition. for example, a mixture of 94.5% ethyl alcohol and 4.5% water by volume.
Maximum boiling azeotrope
These are the binary mixtures whose boiling point is more than either of the two components. The solutions that show large negative deviation from Raoult’s law form maximum boiling azeotrope at a specific composition, e.g., a mixture of 68% HNO3 and 32% H2O by mass.
Electro chemistry Videos :-
Video-1
Video -2
Video-3
Colligative properties:
Certain properties of solutions depend only on the number of particles of the solute (molecules or ions) and do not depend on the nature of solute, such properties are called colligative properties. These are:
(i) Relative lowering of vapour pressure,
(ii) Depression in freezing point,
(iii) Elevation of boiling point,
(iv) Osmotic pressure of the solution.
1. Relative lowering of vapour pressure:
On adding non volatile solute in solvent, vapour pressure of the resulting solution decreases because non volatile solute covers the surface of solvent so escaping tendency of solvent molecules decreases hence vapor pressure of solvent decreases.
If po is the vapor pressure of pure solvent and P is vapour pressure of solution then
(po – p) is the Lowering in vapor pressure and (Po – P) / P is known as Relative lowering in vapour pressure.
The Relative lowering of vapour pressure is the ratio of lowering of vapour pressure and vapour pressure of pure solvent which is equal to the mole fraction of solute.
(Po – P) / Po = XB
2. Elevation of the boiling point:
Boiling point is the temperature at which the vapor pressure of a liquid becomes equal to the atmospheric pressure. As discussed in above topic, on addition of non volatile solute vapor pressure of solution decreases hence more heat is required to make the vapour pressure equal to atmospheric pressure Hence solution boils at higher temperature than pure solvent. this increment in boiling point is known as Elevation in boiling point.
If Tbo is the boiling point of Pure solvent and Tb is the boiling point of solution. the
Elevation in Boiling point is = Tb – Tbo

Depression in freezing point
Depression of freezing point: The lowering of vapour pressure of solution causes a lowering of freezing point compared to that of pure solvent .The difference in freezing point of the pure solvent ( Tfo ) and solution (Tf ) is called the depression in freezing point. dTf = Tfo – Tf
For a dilute solution depression in freezing point is a colligative property because it is directly proportional to molal concentration (m) of solute.

Osmosis
The process in which there is net flow of solvent to the solution by a semipermeable membrane is called osmosis.
Osmotic Pressure
Osmotic pressure: The extra pressure that is applied to stop the flow of solvent to solution across a semipermeable membrane is called osmotic pressure of the solution.
According to Van’t Hoff Charles law and Van’t Hoff Boyles law:- For dilute solution, osmotic pressure is proportional to the molar concentration (C) of the solution at a given temperature T
Thus
π = CRT
as π is the osmotic pressure and R is the gas constant value is 0.08206 L atm Mol-1 K-1.

Reverse osmosis:
The direction of osmosis can be reversed, if a pressure larger than the osmotic pressure is applied to the solution side. Now the pure solvent flows out of the solution through the semipermeable membrane. This phenomenon is called reverse osmosis. Reverse osmosis is used in desalination of ocean water

Types of Solution
1. Two solutions having same concentration or osmotic pressure at a given temperature are called isotonic solution.
2. If a solution has more osmotic pressure than other solution it is called hypertonic solution. or If the concentration of solution is higher than that of cell then solution is called hypertonic solution.
3. If a solution has less osmotic pressure than other solution it is called hypotonic solution. Or If the concentration of solution is less than that of cell then solution is called hypertonic solution
Abnormal Molar mass.
Molar mass that is either lower or higher than expected or normal
molar mass is called as abnormal molar mass.
Molar mass that is either lower or higher than expected or normal molar mass is called as abnormal molar mass. The molecular mass becomes abnormal due to association or dissociation of solute in given solvent. It can be calculated as normal molecular mass using the Van’t Hoff factor (i).
Van’t Hoff Factor
It is the ratio of normal molecular mass to observed molecular mass. It is denoted as i
i = normal molecular mass / observed molecular mass. Or
i = no. of particles after association or dissociation / no. of particles before dissociation or association.
(a) A non-volatile solute undergoes dissociation, then i > 1.
(b) A non-volatile solute undergoes association, then i < 1.
(c) For Non electrolyte Solute i = 1
Degree of Dissociation
Degree of dissociation = (i-1) / (n-1) Where i = Van’t Hoff factor, n = No. of particles after dissociation,
Degree Of Association
Degree Of Association = (i-1) / (1/n – 1) Where i = van,t Hoff Factor, n is the number of particles befor association.
